Walter Isaacson in A Spur to the Biotech Century Ahead (WSJ oped)
The coronavirus plague will hasten our transition to the third great innovation revolution of modern times. These revolutions arose from the discovery, beginning just over a century ago, of the three fundamental kernels of our existence: the atom, the bit and the gene.
How’s that for some hype?
I was pretty excited to read this book and it didn’t disappoint. Isaacson is an amazing writer (I loved his Steve Jobs bio, and have just received the Da Vinci one…) and it’s pretty clear this is the definitive account on the CRISPR story. Bacteria have been fighting off viruses for more than a billion years by effectively storing pieces of attacking viruses’ DNA in their own DNA, transferring some immunity to future generations. Now it’s our turn to do the same.
This was also a smooth high-level intro to a bunch of tangential topics I know nothing about e.g:
- Curiosity-driven vs applied science
- “Science advancing by publicity” and those who don’t do publicity getting largely left behind in praise / recognition
- The brutal races between labs to be the first to publish, get patents granted, and form biotech companies around these patents
- The “RNA world hypothesis”
- Efforts from scientists to regulate their own field (similarly to what’s happening in AI) and a roundup of bioethics arguments
My rough highlights follow, enjoy! 🐛
Curiosity-driven science
On the importance of funding research even if it doesn’t yet have known applications we can foresee:
Research about surface-state physics eventually led to the transistor and microchip. Likewise, studies of an astonishing method that bacteria use to fight off viruses eventually led to a gene-editing tool and techniques that humans can use in their own struggle against viruses.
- Vannevar Bush “linear model of innovation”, led to the NSF
- There was some of this in case of CRISPR (Mojica doing purely curiosity-driven science studying archaea (single cell organisms like bacteria, thriving in salt ponds) but also applied science pushing the field forward from Danisco (yogurt makers)
Mendel & Heredity
The paths that led Watson and Crick to the discovery of DNA’s structure were pioneered a century earlier, in the 1850s, when the English naturalist Charles Darwin published On the Origin of Species and Gregor Mendel, an underemployed priest in Brno (now part of the Czech Republic), began breeding peas in the garden of his abbey.
On Mendel’s breeding of plants with different traits together (e.g white and violet)
What his experiments showed was momentous, given what Darwin was writing at the time. There was no blending of traits. Tall plants cross-bred with short ones did not produce medium-size offspring, nor did purple-flowered plants cross-bred with white-flowered ones produce some pale mauve hue. Instead, all the offspring of a tall and a short plant were tall. The offspring from purple flowers crossbred with white flowers produced only purple flowers. Mendel called these the dominant traits; the ones that did not prevail he called recessive.
- Science advances by publicity, and Mendel was a recluse. His findings were largely ignored until they were rediscovered.
- The findings of Mendel and these subsequent scientists led to the concept of a unit of heredity, what a Danish botanist named Wilhelm Johannsen in1905 dubbed a “gene.” There was, apparently, some molecule that encoded bits of hereditary information.
Human Genome Project
- Started by Watson in 1986, formal launch in 1990. Personal motivation with his son Rufus who was schizophrenic
- Public group with Francis Collins (later NIH) and Eric Lander (mathematician, Rhode scholar, geneticist at MIT)
- vs Private sector group led by Craig Venter (US navy in Vietnam then biotech entrepreneur). Formed Celera to commercialise efforts. Ultimately won
- Tensions between groups but Bill Clinton pushed for them to show a united front. $3b public money
- George Church skeptical on ROI. We mapped but didn’t come up with any treatments
Cold Spring Harbor Laboratory
- Founded in 1890, large campus in Long Island
- Important research there, but also eugenics from 1904 to 1939
- Watson director 1968-2007
- Ousted for repeated eugenics-leaning comments and talking about genetic inferiority of Africans…
RNA
Ribonucleic acid (RNA) is an important biological macromolecule that is present in all biological cells. It is principally involved in the synthesis of proteins, carrying the messenger instructions from DNA, which itself contains the genetic instructions required for the development and maintenance of life
- Messenger molecule that carries instructions from the DNA (that is nested in the nucleus of the cells) to the manufacturing region of the cell
- Facilitates proper sequence of amino acids to make specified proteins
These proteins come in many types. Fibrous proteins, for example, form structures such as bones, tissues, muscles, hair, fingernails, tendons, and skin cells. Membrane proteins relay signals within cells. Above all is the most fascinating type of proteins: enzymes. They serve as catalysts. They spark and accelerate and modulate the chemical reactions in all living things. Almost every action that takes place in a cell needs to be catalyzed by an enzyme.
- Ribozymes (or catalytic RNA): RNA molecules which can both store genetic information and act as a catalyst for reactions
This had pretty cool implications. If some RNA molecules could store genetic information and also act as a catalyst to spur chemical reactions, they might be more fundamental to the origins of life than DNA, which cannot naturally replicate themselves without the presence of proteins to serve as a catalyst.
- An intron (for intragenic region) is any nucleotide sequence within a gene that is removed by RNA splicing during maturation of the final RNA product.
Doudna’s PhD
- Her school councellor (before college): “girls don’t do science”…
- Under Jack Szostak at Harvard’s department of Molecular Bio
- Initially studying how to edit DNA of yeast
- For PhD: studying if ribozymes have “the chemical chops to copy themselves”
- Very few people were studying RNA at the time “Never do something that 1000 other people are doing” – Szostak
- The RNA world hypothesis. That RNA was able to replicate itself in early days of Earth, and then DNA and proteins appeared.
Post-doc in biochemistry
- Biochemistry: how chemical molecules in living cells behave
- Structural biology: lower-level, discover 3D shape of molecule (what Rosalind Franklin had done for structure of DNA, using X-ray crystallography)
- Doudna descided to go do post-doc in Tom Cech’s lab, best RNA biochemistry lab (Uni of Colorado in Boulder)
Doudna’s mission was to map the intron that Cech had discovered could be a self-splicing piece of RNA, showing all of its atoms, bonds, and shapes. If she succeeded in figuring out its three-dimensional structure, that would help show how its twists and folds could bring the right atoms together to cause chemical reactions and allow the snippet of RNA to replicate itself.
- Crystallising RNA
- Got some help from Yale lab to do cryocooling, then she took a job at Yale
She and Cate, along with their lab colleagues, were able to determine the location of every atom in a self-splicing RNA molecule. Specifically, they showed how the structure of a key domain of the molecule allowed RNA to pack helices together to create its three-dimensional shape. A cluster of metal ions in that domain formed a core around which the structure folded. Just as the double-helix structure of DNA revealed how it could store and transmit genetic information, the structure discovered by Doudna and her team explained how the RNA could be an enzyme and was able to slice, splice, and replicate itself.
Viruses & RNA interference
- Interest in how the RNA in some viruses allow them to hijack the protein-making machinery of cells
- Example: SARS-Cov-1 composed of RNA instead of DNA
- RNA interference: small molecules that interfere with the messenger RNAs
- Works by deploying an enzyme “Dicer”
- Doudner set out to disover the structure of Dicer
- Reengineering Dicer enabled eg turning off certain genes which was very useful for research
- Throughout history some organisms have used RNA interference to fight off viruses… but never humans (until…!)
Clustered Repeats
- We had discovered repeated sequences in the DNA of E.Coli but didn’t know what it was for
- Francisco Mojica, a grad student at university of Alicante, was studying archaea (single cell organisms like bacteria, thriving in salt ponds)
- Archaea and bacteria have small amount of genetic material so he knew they couldn’t afford to waste it. He kept pushing although v little funding
- Came up with name CRIPR: “Clustered Regularly Interspersed short Palindromic Repeats”
- In most organisms that had CRISPRs, the repeated sequences were flanked by one of these genes, which encoded directions for making an enzyme. He named these “CRISPR-associated,” or Cas, enzymes.
- Bacteria immune if they have the the CRIPR sequence of a virus. Defence/immune system.
- He couldn’t get published in Nature and took 2 years to get published elsewhere…!
- Started the CRIPR wave
DNA modification not RNA interference
- Jilian Banfield microbiologist who introduced Doudna to CRISPR. She thought it worked by RNA interference
- At that time no one had isolated the molecular components of the CRIPR system
By 2008, scientists had discovered a handful of enzymes produced by genes that are adjacent to the CRISPR sequences in a bacteria’s DNA. These CRISPR-associated (Cas) enzymes enable the system to cut and paste new memories of viruses that attack the bacteria. They also create short segments of RNA, known as CRISPR RNA (crRNA), that can guide a scissors-like enzyme to a dangerous virus and cut up its genetic material. Presto! That’s how the wily bacteria create an adaptive immune system!
- Danisco Danish yogurt makers. Making starter cultures and great threat of virus to the market ($40b/year market). Removed Cas9 gene and showed you lost resistance
- Danisco started using CRISPR to vaccinate its bacterial strains in 2005
- First CRISPR conference 2008
- That year Marraffini and Sontheimer showed that the target of the CRIPR system was DNA, it didn’t work through RNA interference as previously thought
- They were the first to suggest CRISPR-Cas for gene-editing tools
CRISPR Companies / Applications
Caribou Biosciences
- Started by Rachel Haurwitz from Doudna lab in 2011, with Doudna as advisor.
- Exclusive License of Doudna tech from Berkeley
- Therapies in pipeline:

- The name is a cut-and-splice mash-up of “Cas” and “ribonucleotides,” which are the building blocks of RNA and DNA.
CRISPR Therapeutics
- Emmanuelle Charpentier and Rodger Novak
Editas
- Church, Zhang, and Doudna but she bailed early
Mammoth Biosciences
- Doudna co-founder
- Developing tests using CRISPR
Emmanuelle Charpentier
- Had honed in on enzyme called Cas9
- If deactivated the CRISPR system no longer cuts up the invading virus
CRISPR RNAs, known as crRNAs. These are the small snippets of RNA that contain some genetic coding from a virus that had attacked the bacteria in the past. This crRNA guides the Cas enzymes to attack that virus when it tries to invade again. These two elements are the core of the CRISPR system: a small snippet of RNA that acts as a guide and an enzyme that acts as scissors.
- trans-activating CRISPR RNA, tracrRNA
- First, it facilitates the making of the crRNA, the sequence that carries the memory of a virus that previously attacked the bacteria. Then it serves as a handle to latch on to the invading virus so that the crRNA can target the right spot for the Cas9 enzyme to chop.
- Charpentier worked on proving this
- Doudna-Charpentier focus on understanding mechanisms of CRISPR-Cas9
- Paper published in Nature in 2012
The experiments looked at the CRISPR-Cas9 system of Streptococcus pyogenes, a common bacteria that can cause strep throat. Like all bacteria, it is a single-cell organism without a nucleus. But the paper suggested that the CRISPR-Cas9 system could be useful for gene editing in humans. Charpentier thought that would prompt some questions. “I was thinking that the reviewers would ask if there was any evidence that it worked in human cells,” she recalls. “But they never raised that, even after the conclusion I wrote saying that it would be an alternative to existing gene-editing methods”
Competition
- Virginjijus Siksyns
- Doudna got the abstract from his paper and it pushed her to publish hers
- George Church. 2 undergrads in 2 years at Duke, then skipped into PhD and started publishing. Then went to Harvard under Gilbert working on DNA sequencing. Clashing approach to Lander
- Woolly mammoth restoration project
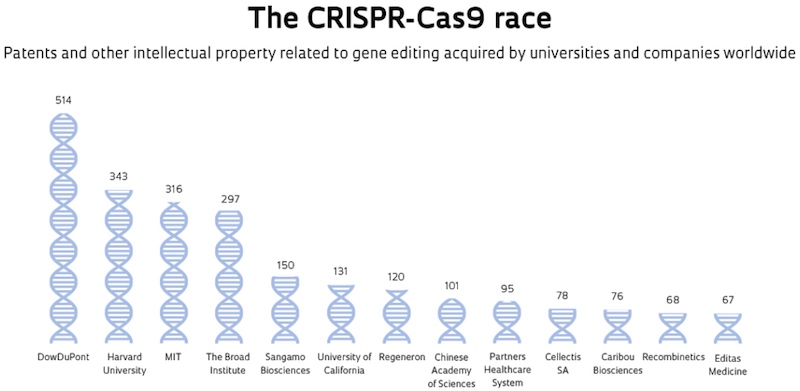
Patent wars
- CRISPR system that worked in single-cell organisms (bacteria and archae) – would they work in multi-cell organisms? Would getting it to go from single-cell to multi-cell be considered major invention?
- If so credit to Broad Institute (Lander, Zhang) if not more credit to Doudna, Charpentier
- Church also got there but after Broad
In 1873, for example, the French biologist Louis Pasteur was awarded the first known patent for a microorganism: a method for making “yeast free from organic germs of disease.” Thus we have pasteurized milk, juice, and wine.
Stanford, for example, made $225 million in twenty-five years by granting hundreds of biotech companies non-exclusive licenses to the Cohen-Boyer patents (recombinant DNA for gene editing, first patent of bio process)
- Supreme court: “a live, human-made micro-organism is patentable” if it is “a product of human ingenuity.”
- Bayh-Dole Act, which made it easier for universities to benefit from patents, even if the research was funded by the government.
Gene editing
- 1972 first DNA splicing (one virus’s DNA into another). Cloning. Birth of biotech field
- Gene therapy: involved delivering into the patient’s cells some DNA that had been engineered to counteract the faulty gene that caused the disease.
- First trial 1990
- 1999 young man died in a trial, developed massive immune response
- 2000 triggered cancer-causing gene leading to leukemia in 5 patients
Editing source DNA
- “Double-strand break” using a specific enzyme (nucleases), then find a guide that would navigate the enzyme to the right place
- In 2000 found Fokl enzyme to do this
- Zync Fingered Nucleases (ZFN) and Transcription Activator-like Efflector Nucleases (TALENs) techniques but then CRISPR came along and the guide was not a protein but a snippet of RNA (much easier to modify, with other techniques you had to find a new protein based on where to target)
Feng Zhang
- Broad Institute (MIT, Harvard). Into computers as a kid. Under Church then Lander
- What Doudna & Charpentier had done was “Biochemistry in a test tube” (still single-cell organisms) and he made it a gene-editing paper, testing with real cells
- Was developing the prerequisites in parallel but was behind on understanding tracrRNA
- Beat Doudna to the patent application, dropped Marraffini from his
- Broad patent for CRISPR for gene editing in humans, Berkeley/Doudna for lower-level CRISPR methods, both valid
- In Britain, China, Japan, Australia, NZ, Mexico, Doudna has the main patent
- They probably should have united: Don’t fight over divvying up the proceeds until you finish robbing the stagecoach. (as Kilby of Texas Instruments and Robert Noyce of Intel did for microchip IP)
Therapies
Sickle cell anemia (blood disorder)
- Disease caused by a mutation in a single letter of more than 3b base pairs of a person’s DNA, which causes a kink in the hemoglobin protein
- This protein contorts red blood cells and causes them to clump
- Oxygen can’t get to tissues & organs, severe pain, in most cases death by 50
- Affects 4M+ people worldwide, 80% in sub-Saharan Africa
- First CRISPR therapy for Victoria Gray in the US. Trial led by CRIPR Therapeutics
- She was injected with her own (edited) stem-cells, and it worked, she started producing “fetal” hemoglobin, the healthy kind
- The gene in question helps with malaria immunity however. An example of the complexity – genes playing multiple roles, and have evolutionary reasons for existing
Cancer
- Several trials in place to disable a gene that produces the PD-1 protein which stops T-cell immune responses, eg in the case of lung-cancer
- Promising future work on cancer:
a gene known as P53 encodes for a protein that suppresses the growth of cancerous tumors. It helps the body respond to damaged DNA and prevents cancerous cells from dividing. Humans tend to have one copy of this gene, and cancers proliferate if something goes wrong with it. Elephants have twenty copies of this gene, and they almost never get cancer.
Congenital Blindness (present from birth)
- “In-vivo” treatment, because you can’t extract eye cells
- Treating “Leber congenital amaurosis” a common cause of childhood blindness
Future
- Tay-Sachs
- Huntington’s
- Mutated copy of gene PCSK9 which reduces “bad” cholesterol and risk of coronary heart disease
Biohacking
- Josiah Zayner
- Profiled in Netflix series “Unnatural selection”
- Running “The Odin” to democratise access to these tools…
[He] launched into a pitch about a do-it-yourself “frog genetic engineering kit” that he made in his garage. Available online for $299, it allowed users to cause a frog’s muscles to double in size in a month by injecting CRISPR-edited DNA that turned off the gene that produces myostatin, a protein that inhibits muscle growth once an animal has reached its mature size.

- (He has a pretty entertaining post on his blog called “That time I ate a weed brownie on a private island Peter Thiel flew me to”)
Anti-CRISPRs
- At one point military was single largest funder of (anti-)CRISPR research to help avoid bioweapons (eg mice could be infected with lung cancer via CRISPR)
- Discovered by Joe Bondy-Denomy
- Viruses can infiltrate the bacteria’s DNA with a little sequence that sabotaged their CRISPR system
- Now more than 50 anti-CRISPR proteins discovered
Public Policy
- David Baltimore, organised the first Asilomar conference on regulating gene editing in 1975 (with Paul Berg)
- IVF – first test-tube baby Louise Brown born in 1978
- Europe no germline editing allowed (Oviedo Convention) except UK and Germany.
- Nuffield Council (leading bioethics body in the UK) very “bioliberal”
- US much more permissive.
- Russia permissive – Putin talking about CRISPR for embryos in 2017
- Jesse Gelsinger tragedy – gene therapy for a liver disease which usually kills babies
- He was surviving on low-protein diet and 32 pills per day
- Massive immune response and died in September 1999
- Bioethics conference in Napa Valley in Jan 2015 organised by Doudna
- Difference with 1975 conference is now there is a big biotech industry. So harder to put the cat back in the bag (before was just government-backed)
- Baltimore against the “fairness” argument of cost prohibitive treatments. Points to computers democratisation says same will happen here
- Agreement that using CRISPR tools for non-inheritable gene editing in somatic cells (which are not transmitted to descendents)
- But they recommended a temporary halt on germline editing
- Chinese embryo work, April 2015
- Led by He Jiankui. Cutting out a mutated gene in embryos that leads to sickle-cell anemia
- Very controversial, done in isolation (even to China regulators), and there were alternative therapies to ways to achieve same outcome
- Red lines
- The germline
- Treatment vs enhancement
- Character. Miles Davis with sickle cell. FDR with polio
- Sports
For example, almost every champion runner has what is known as the R allele of the ACTN3 gene. It produces a protein that builds fast-twitch muscle fibers, and it is also associated with improving strength and recovery from muscle injury.
- Rawls: veil of ignorance leading to justification of genetic engineering only if it doesn’t increase inequality
- Nozick (also at Harvard): “social rules should arise through the voluntary choice of individuals”. Libertarian view. “Genetic supermarket”.
- Michael Sandel argument against “playing God”
If we humans find ways to rig the natural lottery and engineer the genetic endowments of our children, we will be less likely to view our traits as gifts that we accept. That would undermine the empathy that comes from our sense of “there but for the grace of God go I” toward our fellow humans who are less lucky. “What the drive to mastery misses and may even destroy is an appreciation of the gifted character of human powers and achievements,” Sandel writes. “To acknowledge the giftedness of life is to recognize that our talents and powers are not wholly our own doing.”
COVID
- For the SARS-CoV-2 coronavirus, the human receptor is a protein known as ACE2.
Testing SNAFU
January 2020:
Within two weeks, Greninger had a working test, which, under normal regulations, they could use in their own hospital system. But then HHS Secretary Azar [FDA] issued his emergency declaration, which made regulations more strict. So Greninger submitted a formal application to the FDA for an “Emergency Use Authorization.” It took him close to one hundred hours to fill out all of the forms. Then came an astonishing bureaucratic snafu. He got a response from the FDA on February 20 informing him that, in addition to sending his application electronically, he had to mail in a printed copy along with a copy burned onto a compact disc (remember what those were?) to FDA headquarters in Maryland. In an email he wrote to a friend that day describing the FDA’s bizarre approach, Greninger vented, “Repeat after me, emergency.”
How PCR tests work
… a long swab is inserted into the back of a patient’s nasal passage. The lab uses some of the chemical mixtures in the kit to extract any RNA that is in the mucus. The RNA is then “reverse-transcribed” to turn it into DNA. The DNA strands are amplified into millions of copies using a well-known process called a polymerase chain reaction (PCR), which most college biology students learn how to do.
CRISPR-based testing
- Doudna’s Innovative Genomics Institute. Allowed for non-exclusive no-fee licensing of all their COVID work. Initially produced tests
- Suggested using CRISPR to detect the RNA of the coronavirus
- In 2017-2018, CRISPR test called DETECTR for STD called HPV. Mammoth biosciences born to commercialise these testing techniques
- Rival similar technique from Zheng / the Broad called SHERLOCK. Showed detection of specific strains of Zika and Dengue first. Sherlock biosciences born from it
- Covid tests based on CRISPR now available from Mammoth and Sherlock. As fast as antigen test but more accurate
- Antigen tests detect the presence of proteins that exist on the surface of the virus, most accurate only when the patient has become highly infectious to others
- CRISPR-based tests can detect presence of the RNA of a virus as soon as a person has been infected
Vaccines
- Traditional vaccine – kick the immune system into gear by introducing a small amount of a virus or other patogen
- Genetic vaccines. Deliver a piece of genetic coding that will guide human cells to produce, on their own, components of the virus, which stimulate the immune system
- Oxford uni. Reengineered a safe virus (an adenovirus that causes flu in chimpanzees) by editing into it the gene to make the spike protein of the coronavirus. Led to Ox-Astra vaccine. Initial funding from Bill & Melinda Gates foundation
- J&J does something similar with a human adenovirus
- DNA vaccines
- None had been approved before Covid
- Inovio Pharmaceuticals and others had created a circle of DNA that coded for parts of the coronavirus spike protein
- Big challenge is delivery – how to get it into the nucleus of the cell.
- Marson & Wilson (Berkeley, UCSF) put together a “shuttle” that could get the DNA vaccine into cells using a Cas9 protein, guide RNA, and a “nuclear localization signal”
- RNA vaccines
- The mRNA instructs cells to make part of the spike protein that is on the surface of a coronavirus
- Advantage is doesn’t need to go to the nucleus of the cell, works in outer region (cytoplasm)
- BioNTech launched Project Lightspeed
- Moderna. Funded by US government, Fauci said “whatever it costs, don’t worry about it”
- Moderna created required RNA sequences in 2 days, 38 days later started early stage trials
- They had been working for 10 years on perfect lipid nanoparticles to carry the molecules into a human cell. Doesn’t have to be stored at extremely low temperatures like BioNTech one
CARVER and PAC-MAN
- Cameron Myhrvold (son of Nathan) & Stanley Qi respectively
- Working on using CRISPR-Cas13 to do for humans what it does for bacteria: target a dangerous virus and chop it up
- Would work more efficiently than having to trigger the body’s erratic immune response
- Still in early stages
Nobel
- 2020 Chemistry Nobel for Doudna & Charpentier 🎉
Good summary:



[…] Alfred Russel Wallace coming onto the same theory and pushed Darwin to publish his work (competition spurring publications once again… lots of that in the CRISPR story) […]
LikeLike
[…] Covered in my review of The Code Breaker […]
LikeLike